Struggling with outsourced clinical trials?
You’re not alone.
Clinical trials often face critical challenges that affect their success and efficiency.
Inefficient or flawed study design, leading to complications in data collection
Inaccurate, incomplete, or inconsistent data
Invalid results due to poor data quality
Insufficient oversight and lack of traceable documentation
Inconsistent data storage leading to potential compliance risks
We support you with our expertise in avoiding these challenges.

Our Services for Trial Oversight
Efficient study oversight – clear, flexible, reliable.
Whether it’s strategic guidance, document review or real-time oversight – we bring structure to your processes and confidence to your decisions.

Clinical trial oversight
Our Octovis dashboard gives you clarity before things get critical: Early warning indicators, clean visualizations, and audit-ready summaries – true oversight made simple, for traceable results and high-quality documentation.
Review & Quality Assurance
We review what matters: SOPs, Data Management Plan, TLFs, and Clinical Study Reports – for highest data quality.
Consulting & Strategic Support
From CRO selection and protocol and e-CRF design to CTMS implementation – we offer expert input, tailored to your needs.
Collaboration that fits your workflow
Whether you need targeted support, full-scale involvement, or embedded project work – we adapt to your setup.
Strategic planning from day one
Included services:
- Expert consulting on study design and protocol development
- Structured risk analysis and scenario planning
- SOP review and harmonization across Sponsor and CRO
- Support in selecting the right EDC system for your needs
- SDTM conversion of your trial data by our experienced team
Biostatistics with clear focus
- Expert consulting on endpoints and overall statistical planning
- Review and optimization of the Statistical Analysis Plan (SAP) and randomization schemes
- Support with data analyses, output programming, and statistical tables/figures/listings (TFLs)
- Guidance through interpretation and documentation of statistical results in the Clinical Study Report (CSR)

Oversight that enables action
- Tailored configuration of Octovis, our clinical trial oversight platform
- Visualization of study progress, risk signals, and key priorities
- Dynamic updates to the oversight view as new challenges emerge
- Integrated KPI tracking, data management, and monitoring support
- Built-in audit trail functionality to document and track oversight activities for compliance
- Transparent documentation of oversight processes – ensuring traceability, accountability, and inspection readiness
Smart Data Management Oversight
- Review of CRF and Annotated CRF
- Data Management Plan (DMP) review and optimization for project fit
- SAE/AE/MedDRA reconciliation and issue resolution
- Planning and execution of User Acceptance Testing (UAT) – including
- Medical review support with subject-level drilldowns
- Quality and consistency checks for key submission documents
We work the way you work
-as if we were part of your in-house unit.
From protocol development to submission and final archiving, we’re by your side.
We support you with experience, structure, and a sense for what truly matters.
Support that truly fits, because every study is different:
with its own dynamics, challenges, and objectives.
So you can focus on what drives your business forward.
Curious to learn more?
Let’s talk – and find the best path forward for your clinical trials.
Transparency and control –
anytime, anywhere
Key Features:
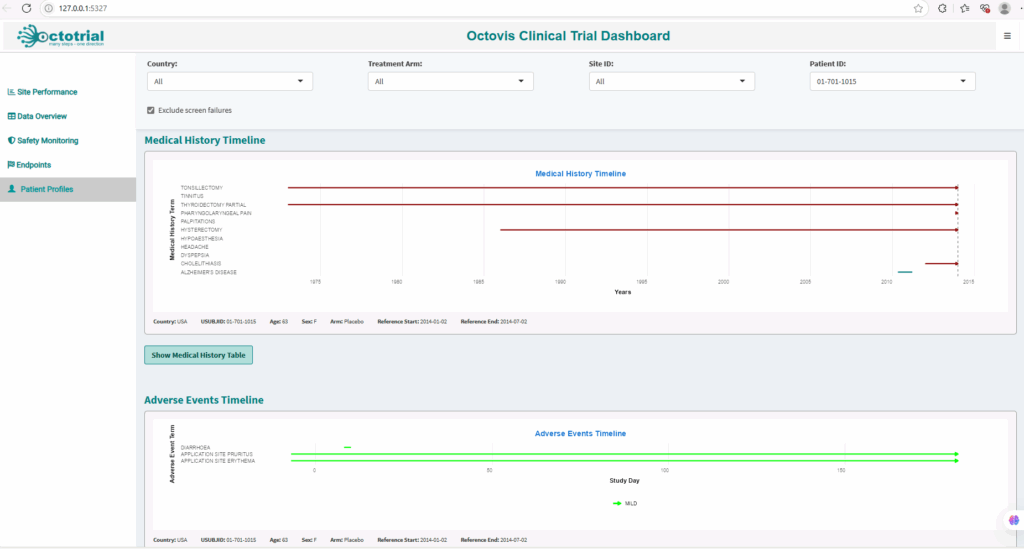
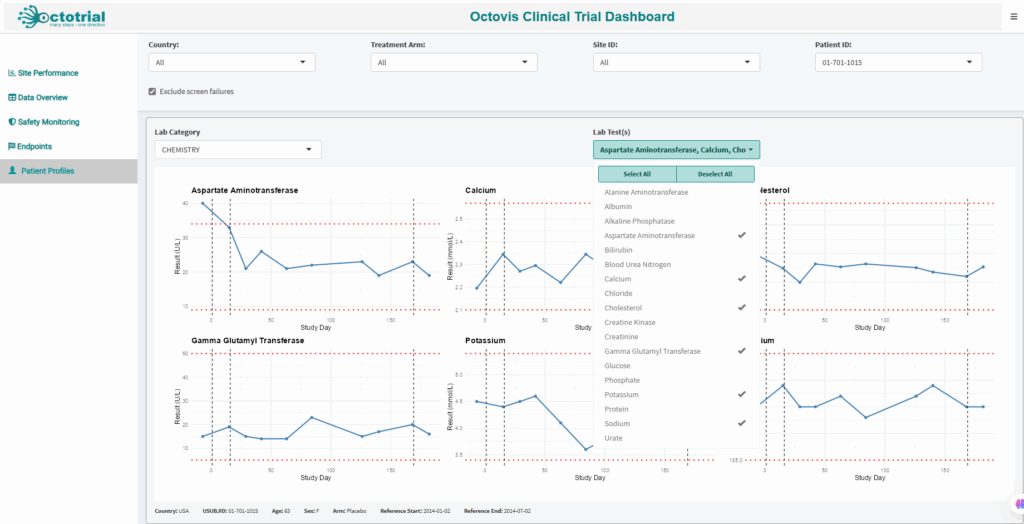
Patient Profiles:
Interactive profiles showing key data such as demographics, treatment history, and adverse events, enabling real-time tracking and safety assessments.Safety Monitoring:
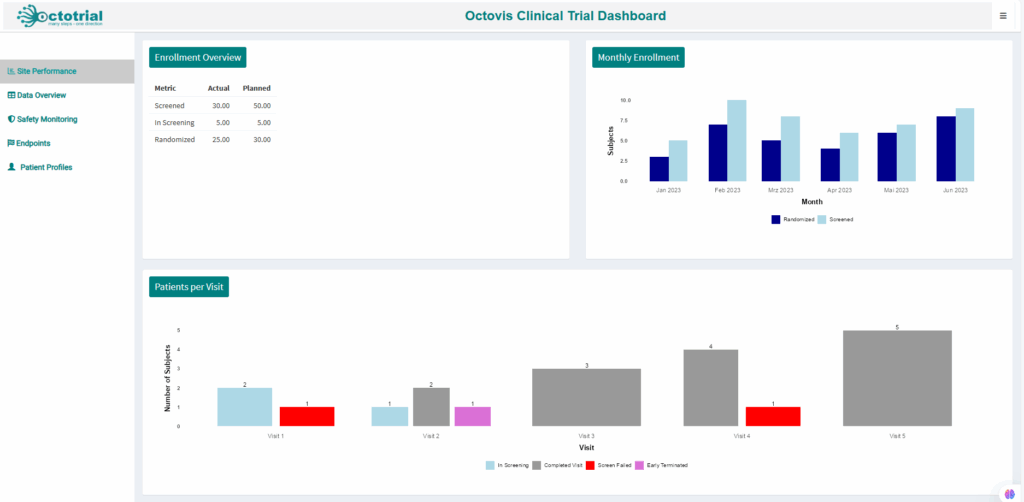
Visualize adverse events and safety trends, with early warning indicators to address potential risks quickly and ensure patient safety.Site Performance:
Track site progress with real-time metrics on recruitment, data quality, and study milestones, allowing you to optimize site performance.Audit Trail Analysis:
Access a comprehensive, audit-ready trail of all activities, ensuring full traceability and regulatory compliance according to ICH E6(R3) GCP guidelines.Study-Specific Plots & Reports:
Fully customizable plots and reports tailored to your study’s specific needs, giving you in-depth insights for smarter decision-making.
About Us

Ortrud Junker-Wolf
Founder// Programming // Biostatistics
Data Scientist specializing in clinical research, with expertise in statistical programming, biostatistics, and data management. I help turn complex, fragmented data into actionable insights, enabling smarter, data-driven decisions. Passionate about making data work for better outcomes in clinical trials. With my work, I ensure that innovative therapies reach patients quickly.

Torsten Blass
Data Scientist // Programming // Data Management
Data Scientist who bridges the gap between clinical needs and technical solutions. With a biochemistry background and passion for statistical programming, I transform client requirements into intuitive visualizations and seamless functionality. I thrive on finding the optimal way to display complex data, maintaining code integrity, and implementing features that make clinical oversight both powerful and accessible.

Claudia Vollmer
Project Manager // Design // Communication
What to expect when
working with us
Structured thinking meets technical precision: You’ll work with someone who speaks your language – and translates it into practical solutions.
Tailored support: No one-size-fits-all system – but a solution adapted to your study design, your team, and your needs.
Reliable communication: Direct contact, clear agreements, dependable delivery.
A love of efficiency: I thrive on clean workflows, elegant visualizations, and small tools that make a big difference.
We look forward to helping you manage your trials with more intelligence, and a little more peace of mind.

Get in touch with us!
We are looking forward to hearing from you!
Please contact us: contact@octotrial.com